Idiosyncratic adverse drug reactions (ADRs) include examples of strong associations between genotypes and drug responses. These reactions:
- i) are potentially life-threatening,
- ii) they cannot be simply predicted from the known pharmacology of the medicinal product or its plasma concentration,
- iii) they are rare, therefore not usually detected in clinical trials during development and
- iv) thus only become apparent when many individuals are exposed to the culprit agent after marketing.
It is therefore recommended to collect DNA for later HLA typing in a drug developmental program to identify possible useful predictive HLA biomarkers. A comprehensive database listing ADRs associated with the HLA alleles and haplotypes is available.
HLA typing should be undertaken by accredited laboratories using DNA based methods. High resolution typing of the following six genes shall be performed: HLA-A, -B, -C, -DR, -DQ and –DP. The nomenclature for HLA alleles is given in below Figure:
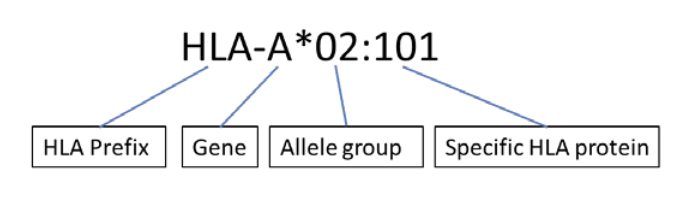
Figure: Nomenclature of HLA alleles
More than 15,000 different HLA alleles have been reported. Interpretation of HLA typing results is complex and it is not always possible to define the type to a single allele. Ideally a technology should be used that allows sequencing of the entire HLA gene, which removes ambiguity of HLA types produced. It also allows the analysis of the HLA haplotypes.
The advent of NGS and the development of computational tools capable of typing the HLA alleles using WGS or WES sequencing data, is likely to become the gold standard approach for HLA typing and current recommendation is to use this when possible.
Pre-emptive genotyping for some HLA alleles is mandatory for use of certain medicinal products either in clinical studies or in practice. These include testing for HLA-B57:01 before starting abacavir, HLA- B15:02 in Asians before initiating carbamazepine treatment and HLA-B*58:01 before starting allopurinol treatment.
These tests have been shown to reduce the incidence of serious hypersensitivity reactions if individuals who test positive are given alternative medications. The development of HLA allele panels with integrated clinical decision support system, pre-emptive genotyping across the HLA region and reducing cost of next generation sequencing promise to deliver cost-effective and efficient HLA testing available at the time of treatment.
Read also: Phenotype and Genotype Correlations

